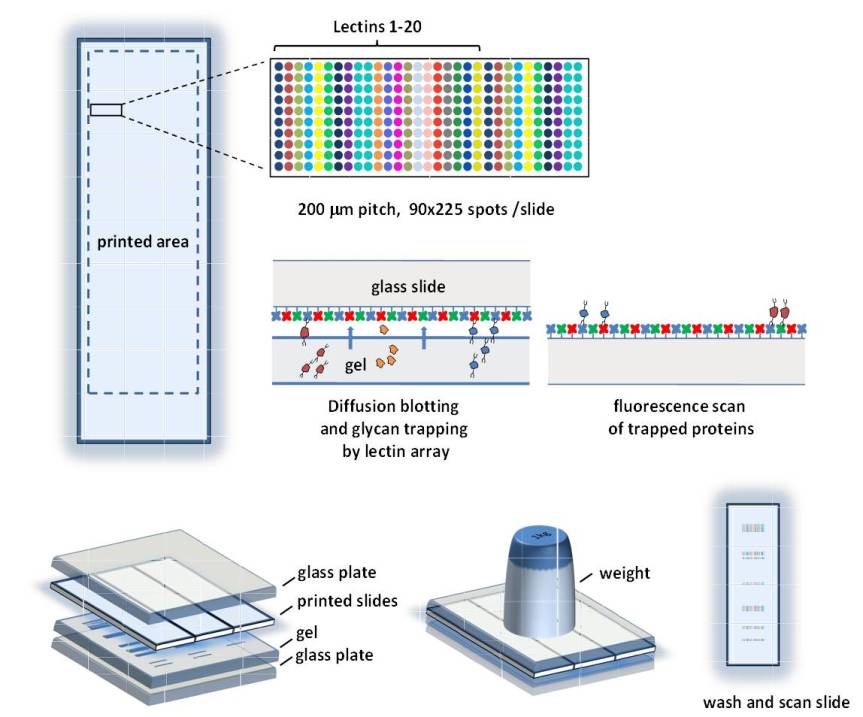
Lectin Array Blot
By combining electrophoretic protein separation with lectin-array-based glycan profiling into a single experiment, we have developed a high-throughput method for the rapid analysis of protein glycosylation in biofluids. Fluorescently tagged proteins are separated by SDS-PAGE and transferred by diffusion to a microscope slide covered with multiple copies of 20 different lectins, where they are trapped by specific carbohydrate protein interactions while retaining their relative locations on the gel. A fluorescence scan of the slide then provides an affinity profile with each of the 20 lectins
containing a wealth of structural information regarding the present glycans. The affinity of the employed lectins toward N-glycans was verified on a glycan array of 76 structures. While current lectin-based methods for glycan analysis provide only a picture of the bulk glycosylation in complex protein mixtures or are focused on a few specific known biomarkers, our array-based glycoproteomics method can be used as a biomarker discovery tool for the qualitative exploration of protein glycosylation in an unbiased fashion.
featured in Chemical and Engineering News
Exosome Glycan Analysis
As cellular-derived vesicles largely maintain the biomolecule composition of their original tissue, exosomes, which are found in nearly all body fluids, have enormous potential as clinical disease markers. A major bottleneck in the development of exosome-based diagnostic assays is the challenging purification of these vesicles; this requires time-consuming and instrument-based procedures. We employed lectin arrays to identify potential lectins as probes for affinity-based isolation of exosomes from the urinary matrix. We found three lectins that showed specific interactions to vesicles and no (or only residual) interaction with matrix proteins. Based on these findings a bead-based method for lectin-based isolation of exosomes from urine was developed as a sample preparation step for exosome-based biomarker research. (Echevarria et al. 2014)

