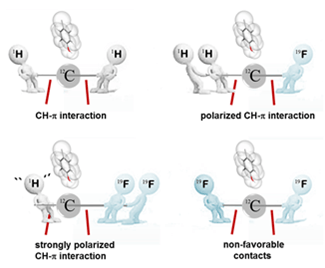
 Together with the groups of Jesus Jimenez-Barbero, CIC bioGUNE, Bilbao, Antonio Franconetti, University of Seville, and Javier Cañada, CIB-CSIC, Madrid we have just published a study in the journal Chemistry, A European Journal titled Fluoroacetamide Moieties as NMR Probes for molecular recognition of GlcNAc-containing sugars: Modulation of the CH-π Stacking Interactions by Different Fluorination Patterns. In the paper we propose the use of the fluoroacetamide and difluoroacetamide moieties as sensitive tags for detecting sugar-protein interactions by simple 1H and/or 19F NMR methods. As selected process, we have chosen the binding of N,N’-diacetyl chitobiose, a ubiquitous disaccharide fragment in glycoproteins, by wheat germ agglutinin (WGA), a model lectin. Using STD-NMR, we experimentally demonstrate that, under solution conditions, the molecule containing the CHF2-CO-NH- moiety is the stronger aromatic binder, followed by the analogue with the CH2F-CO-NH- group and the natural molecule (with the CH3-CO-NH- fragment). In contrast, the molecule with the CF3-CO-NH- isoster displays the weakest intermolecular interaction (one order of magnitude weaker). Since sugar-aromatic CH-π interactions are at the origin of these observations, these results further contribute to the characterization and exploration of these forces and offer an opportunity to use them to unravel complex recognition processes.
Together with the groups of Jesus Jimenez-Barbero, CIC bioGUNE, Bilbao, Antonio Franconetti, University of Seville, and Javier Cañada, CIB-CSIC, Madrid we have just published a study in the journal Chemistry, A European Journal titled Fluoroacetamide Moieties as NMR Probes for molecular recognition of GlcNAc-containing sugars: Modulation of the CH-π Stacking Interactions by Different Fluorination Patterns. In the paper we propose the use of the fluoroacetamide and difluoroacetamide moieties as sensitive tags for detecting sugar-protein interactions by simple 1H and/or 19F NMR methods. As selected process, we have chosen the binding of N,N’-diacetyl chitobiose, a ubiquitous disaccharide fragment in glycoproteins, by wheat germ agglutinin (WGA), a model lectin. Using STD-NMR, we experimentally demonstrate that, under solution conditions, the molecule containing the CHF2-CO-NH- moiety is the stronger aromatic binder, followed by the analogue with the CH2F-CO-NH- group and the natural molecule (with the CH3-CO-NH- fragment). In contrast, the molecule with the CF3-CO-NH- isoster displays the weakest intermolecular interaction (one order of magnitude weaker). Since sugar-aromatic CH-π interactions are at the origin of these observations, these results further contribute to the characterization and exploration of these forces and offer an opportunity to use them to unravel complex recognition processes.

